Author's Guidelines
MANUSCRIPT PREPARATION
The manuscript should be typed, double-spaced on standard-sized-paper A4 (8.5" x 11") with 1" margins on all sides. Times New Roman font 12 pt should be used. The fonts used in the text as well as graphics should be restricted to Times New Roman, Symbol and Zapf Dingbats.
TITLE PAGE
The title page should contain a clear, concise and informative title of the article followed by the names and affiliations of the authors. The affiliation should comprise the department, institution, city, and state (or nation) and should be typed as a footnote to the author's name. The Corresponding Author must indicate his or her complete mailing address, office/cellular phone number, fax number, and email address at the lower left of the Title Page.
ARTICLE STRUCTURE
Abstract
The abstract should not more than 250 words and should contain objectives, material and methods, Results and Conclusions. Reviews and mini reviews also require an abstract. Abstract should be structured. Abstract must be followed by three-six keywords.
Introduction
This should be brief and indicates aim of the study and the essential back ground information. Introduction should clearly state the hypothesis or purpose statement, how and why the purpose or hypothesis was developed and why the author deems it important.
Material and methods
Please provide concise but complete information about the material and the analytical, statistical and experimental procedures used. This part should be as clear as possible to enable other scientists to repeat the research presented. The use of subheadings to divide the text is encouraged. Primary headings should be in BOLD CAPITAL LETTERS. Secondary or subheadings should be in Bold sentence case. Third level subheadings should be in Italicized Sentence Case. In case of animal/human experiments or clinical trials authors must give the details of ethical approval.
Result and Discussion
Data acquired from the research with appropriate statistical analysis described in the methods section should be included in this section. In this part, the same data/ information given in a table must not be repeated in a figure, or vice versa. Tables and Figures should be self-explanatory and it is not acceptable to repeat extensively the numerals from tables into text and give lengthy and unnecessary explanations of the Tables and Figures. Discussion should relate the results to current understanding of the scientific problems being investigated in the field.
Conclusion
This should clearly explain the main conclusions of the work highlighting its importance and relevance.
References
Reference should be cited in the text by author’s last name & year.
Single author: the author's name (without initials) and the year of publication e.g. (John, 2006). Two authors: Last name of both the authors and year of publication should be used i.e. (John and Paul, 2016). Three or more authors: first author's name followed by et al and year of publication i.e. (Mark et al., 2016).
References should be arranged in alphabetical order and further shorted in chronological manner.
Journal
[First author, second author,…... last author]. [Title of Journal]. [Name of journal].[Year];[Volume][(Issue)]: pp (page-page)
Abdillahi HS, Finnie JF, Van Staden J. Anti-inflammatory, antioxidant, anti-tyrosinase and phenolic contents of four Podocarpus species used in traditional medicine in South Africa. Journal of Ethnopharmacology. 2011;136(3):pp 496-503
Reference to a book:
Vyas SP, Khar RK. 2001. Targeted and controlled drug delivery. New Delhi, India: CBS Publisher and Distributor.
Chapter in an edited book:
Chandrasekaran SK, Benson H, Urquhart J. 1978. Methods to achieve controlled drug delivery: The biomedical engineering approach. In: Robinson JR, ed. Sustained and Controlled Release Drug Delivery Systems. New York: Marcel Dekker 557-93.
Electronic Resources:
Webpage with authors
Anchal Sankhyan and Pravin Pawar. 2012. Recent Trends in Niosome as Vesicular Drug Delivery
System. [ONLINE] Available at: http://www.japsonline.com/counter.php=497. [Accessed 08 July 2012].
Abbreviations
Standard abbreviations should be used throughout the manuscript. All nonstandard abbreviations should be kept to a minimum and must be defined in the text following their first use.
Tables & Figures
Tables and figures should not be embedded in the text, but should be included at the end of the manuscript on separate pages. Tables should be created with a word processor and cited consecutively in the text. To ensure the highest print quality, your figures must be submitted in TIF format with minimum 300 dpi or higher resolutions. Captions/legends will be placed below figures and adjusted to 10 font size.
Units and Symbols - The use of the International System of Units (SI) is recommended.
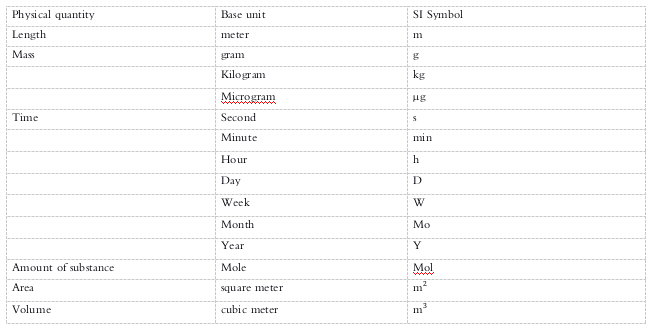

Symbols and abbreviations
Abbreviation must be being wrote in vitro, in vivo, in situ, ex vivo, ad libitum, et al. and so on are two words each and should be written in italics. None of the above is a hyphenated word. All foreign language (other than English) names and words shall be in italics as a general rule. Words such as carrageenan-induced inflammation, paracetamol-induced hepatotoxicity, isoproterenol-induced myocardial necrosis, dose-dependent manner are all hyphenated.
Chemical terminology
The chemical nomenclature used must be in accordance with that used in the Chemical Abstracts.
Biological nomenclature
Names of plants, animals and bacteria should be in italics.
Enzyme nomenclature
The trivial names recommended by the IUPAC-IUB Commission should be used. When the enzyme is the main subject of a paper, its code number and systematic name should be stated at its first citation in the paper.
Spelling
These should be as in the Concise Oxford Dictionary of Current English.
Acknowledgement
All acknowledgments (if any) should be included at the very end of the paper before the references and may include supporting grants, presentations, and so forth.
Ethical Approvals (if applicable)
Patient right to privacy
Patients have a right to privacy thus; all the studies should be conducted with informed consent. No identifying information, should be published in written descriptions, photographs, and pedigrees unless the information is essential for scientific purposes and the patient (or parent or guardian) gives written informed consent for publication. Investigations with human subjects must follow the ethical standards formulated in the Helsinki Declaration of 1964, revised in 2000. All the experiment should be approved by the institutional human experimentation committee or equivalent.
Animal Rights
Animal experiments must follow the ethical standards for the care and use of laboratory animals. Article involved the animal experiments must seek approval from the appropriate Ethical Committee in accordance with "Principles of Laboratory Animal Care" (NIH publication no. 85-23, revised 1985).
Article Processing Charges (APCs)
Authors are required to pay Rp 250.000,00 IDR (Indonesian Authors) or USD 25 (foreign authors) for APCs upon acceptance of manuscript. APCs are applicable only to the accepted articles.
Download Copyright Agreement.docx



